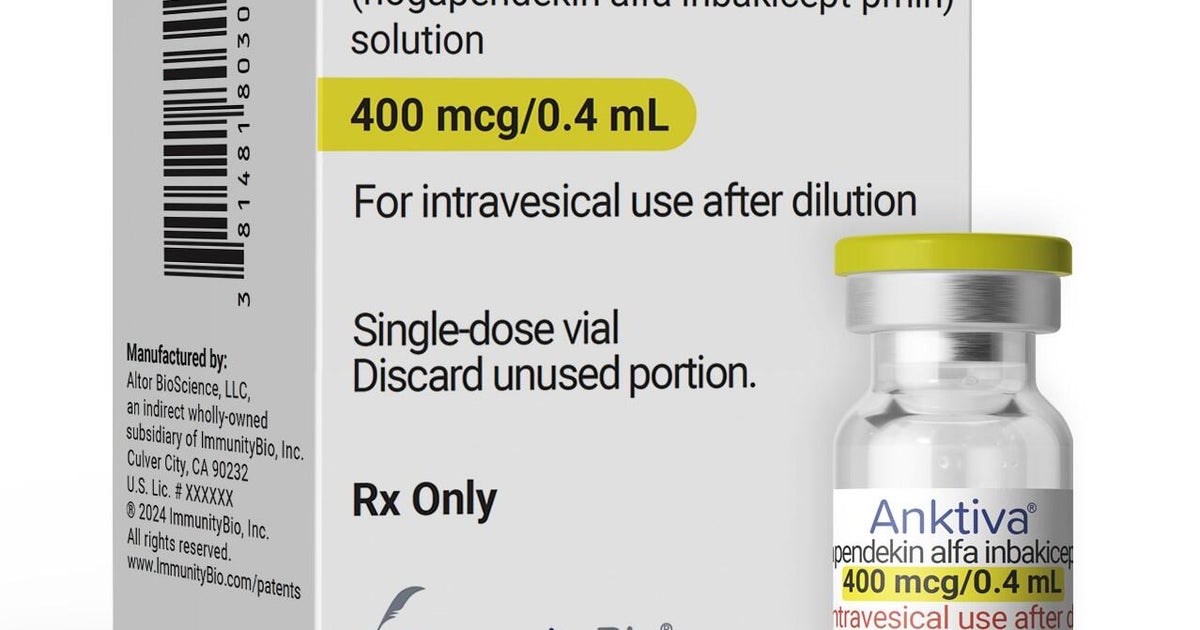
FDA says billionaire's claims about cancer drug Anktiva are misleading
Quick Insights
The Bottom Line
FDA warned billionaire Dr. Patrick Soon-Shiong that his marketing claims about bladder cancer drug Anktiva are misleading.
How This Affects You
If you or a family member considers Anktiva for bladder cancer, FDA warning means promotional claims may overstate the drug's proven benefits or safety profile.
AI Summary
The FDA has issued a warning that biotech billionaire Dr. Patrick Soon-Shiong made misleading claims about Anktiva, his company's bladder cancer drug. Federal health officials posted the warning after determining that Soon-Shiong's statements about the medication did not align with agency standards or approved labeling. The move underscores the FDA's enforcement authority over drug marketing claims, even when made by company founders or high-profile executives. Anktiva received FDA approval, but manufacturers must restrict their promotional statements to what has been clinically demonstrated and approved. The warning signals potential regulatory consequences if Soon-Shiong's company continues making unsupported claims about the drug's efficacy or safety.
What's Being Done
The FDA issued a warning and signaled potential regulatory consequences if Soon-Shiong's company continues making unsupported claims about the drug.
Should this be getting more attention?
You Might Have Missed
Related stories from different sources and perspectives
 Health
HealthTrump and RFK Jr touted leucovorin as a treatment for autism. The FDA quietly walked it back
<p>Outpatient prescriptions for the drug increased 71% after the announcement, but the FDA later approved it only for folate deficiency </p><p>When Donald Trump and Robert F Kennedy Jr stood up at the press conference in September to tout leucovorin, a vitamin B derivative, as a treatment for autism, some neurodevelopmental doctors were shocked – and they braced themselves. There was little evidence to suggest the folinic acid helps with autism, yet there was an immediate flood of parents calling and scheduling visits to talk about the medication.</p><p>“The average parent who maybe wasn’t getting the right information said, ‘Well, to be good parents, we need to try this,’” said William Graf, a professor of pediatrics and neurology at Connecticut Children’s Medical Center. Outpatient leucovorin prescriptions for children ages five to 17 in the US rose by 71% in the weeks following the announcement, new <a href="https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(26)00243...
 Health
HealthFDA backs off stricter tanning bed rules with RFK Jr.'s support
Health officials with the Trump administration have backed away from an effort to more heavily regulate indoor tanning — despite protests from medical groups that warn of the dangers of skin cancer.
 Corporate
CorporateJury finds Elon Musk misled investors during Twitter purchase, absolves him of some fraud claims - AP News
<a href="https://news.google.com/rss/articles/CBMisAFBVV95cUxNS2NWNXFwNUlKbF9WMUZtS0FuZi1Tb3Z0TWhYeExma0ZVWkJvQS1KbTItZkZwd0F6LWRPZzE3TURoR2NVTVMzQjVualBpTVhJMTNvSkw4cDFzVUtwVVBJbW9Ecl9tU3RYRzBCZGhlR3Fvc0FSeHNvN2tmYTFzcVNKbEthbEF1QUVZOEVXakp4NzNvaXhEWkVhdEVrN3RiWEtHUUhjWXNWaUhHOGtaX0ZXSA?oc=5" target="_blank">Jury finds Elon Musk misled investors during Twitter purchase, absolves him of some fraud claims</a> <font color="#6f6f6f">AP News</font>
 Corporate
CorporateJury finds Musk misled investors during Twitter takeover, absolves him of some fraud claims
A jury has found Elon Musk liable for misleading investors by deliberately driving down Twitter's stock price in the tumultuous months leading up to his 2022 acquisition of the social media company for $44 billion. But it absolved him of some fraud allegations, finding that he did not "scheme" to mislead investors.
 Corporate
CorporateJury finds Elon Musk liable for misleading investors during Twitter purchase
A jury has found Elon Musk liable for misleading investors by deliberately driving down Twitter's stock price in the tumultuous months leading up to his 2022 acquisition of the social media company.
 Health
HealthNo Pills or Needles, Just Paper: How Deadly Drugs Are Changing
Lab-made drugs soaked into the pages of letters, books and even legal documents are being smuggled behind bars, killing inmates and frustrating investigators.

Jury orders Meta and Google to pay woman $6 million in social media addiction trial
The verdict marks the end of<strong> </strong>the first-ever jury trial<strong> </strong>over whether tech giants should be held accountable for social media addiction. It may influence the outcome of 2,000 other pending lawsuits.
Did this story change how you see things?
Stories like this only matter when people see them. Help us get verified journalism in front of more eyes.
The Verity Ledger curates verified investigative journalism from trusted sources only.
See our sourcesMost Read This Week
Fentanyl found inside Barbies sold at Missouri discount store, police say
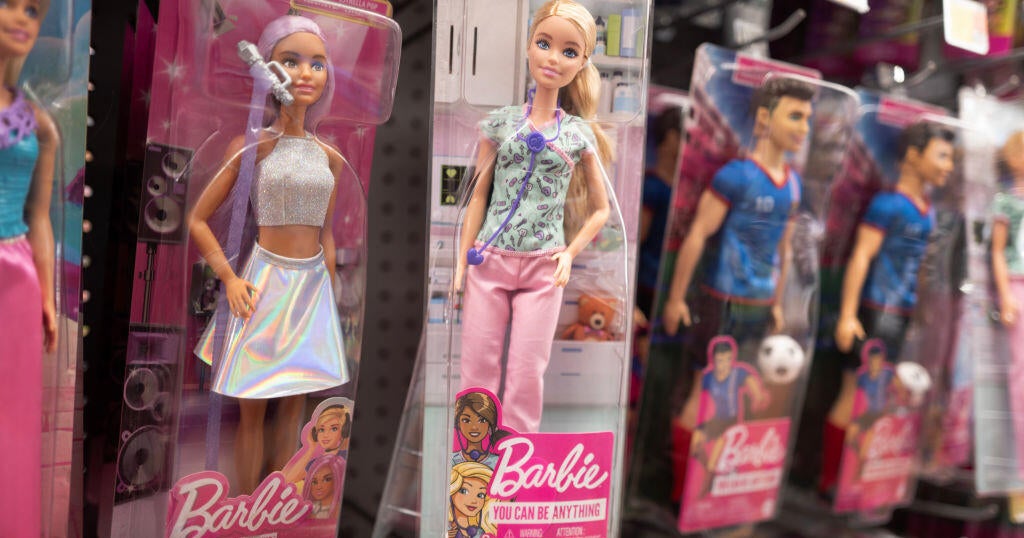
US moves to soften capital rules: ‘Big banks can declare mission accomplished’

Senate deal reached to cap insulin costs

The West's historic snow drought could bring water shortages, wildfires

Pentagon's limits on press access unconstitutional, US judge rules - Reuters